Bringing the woolly mammoth back to life: A conversation on biotech with Harvard Professor George Church
We dip our feet into biotech - Dr. George Church from Harvard Medical School unpacks species revival, the future of gene editing, the business of biotech, and what deeptech investors often overlook
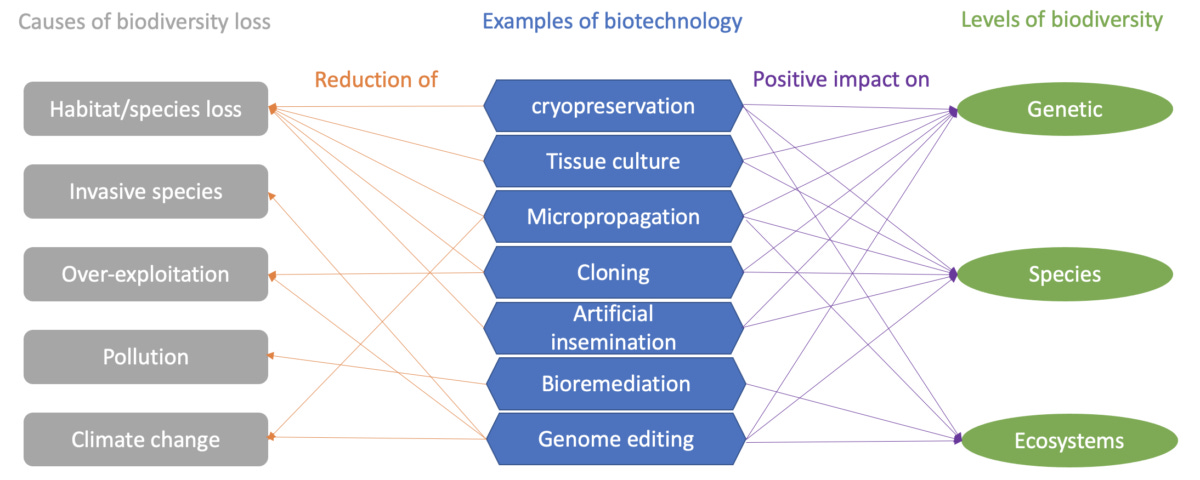
Biotech is a real game-changer for conserving nature and protecting against climate change - it can save endangered species, grow stronger crops with fewer chemicals, clean pollution, track ecosystem health, and vaccinate wildlife.
The framework below from the Alliance for Science showcases some examples of how biotech affects biodiversity in many ways. Take cryopreservation, for instance—freezing genetic material like seeds, sperm, and embryos ensures we can save endangered species and rare plants for the future. Imagine using cryopreservation to take a coral, edit the genome, and make it a supercoral to survive warmer oceans! This is an oversimplification, of course, but you get the general idea
Source: Alliance for Science
This week we want to talk to you about one such application - de-extinction, and the people behind it.
De-extinction?! You’re kidding, right?
Remember Manny and Ellie from the Ice Age movies?
They’re Woolly mammoths, which once inhabited large swathes of Europe, Asia and North America until they went extinct about 4,000 years ago.
Back when mammoths wandered the snowy expanses, their strolling compacted the snow, pushing the cold deep into the earth and keeping the region cooler. Now, without these giant ice-age landscapers, that cooling effect is gone, and the permafrost—ground frozen for at least two years—melts faster. This releases tons of ancient greenhouse gases trapped for millennia. To give you a sense of how serious this is, the carbon released from permafrost melt equals burning the world’s forests two and a half times over. It’s a big deal, and it's not getting the attention it deserves.
Scientists are developing plans to reintroduce the long-extinct woolly mammoth to help tackle severe climate change impacts in tundra and boreal forest areas. Professor George Church from Harvard Medical School shot to fame in 2008 after an NYT article highlighted advancements in decoding genomes and their potential for mammoth revival. This news sparked media sensationalism, with numerous headlines suggesting that a "Jurassic Park: Siberia!" was imminent.
Even beyond the mammoth, many scientists and companies across the globe have dedicated themselves to reviving extinct species such as the dodo, Tasmanian tiger and the passenger pigeon
What does popular media get wrong about this project?
True de-extinction isn't possible yet. Instead, George Church’s startup, Colossal, is working on a hybridization project that aims to create a cold-resistant Asian elephant. This effort focuses on bringing back traits, behaviors, and characteristics of extinct species to help revitalize ecosystems. For example, with the Mammoth project, the intent is not to make perfect copies of extinct Woolly Mammoths, but to focus on the mammoth adaptations needed for Asian elephants to thrive in the cold climate of the Arctic. However, creating hybrid animals, like elephants with mammoth genes, faces many technical challenges. Ultra-precise genome editing is still tough, and our understanding of genetics and development, especially for elephants and birds, has significant gaps.
How does the technology work?
The seeds for de-extinction were sowed in 1984 when the Human Genome Project (HGP) was launched. The HGP profoundly transformed various fields by mapping the entire human genome for the first time in human history, revolutionizing medical diagnostics, and enabling personalized medicine tailored to individual genetic profiles. What does it even mean to map the human genome? Imagine you have a really big book with lots of pages, and each page is filled with words that tell you all about how to build a human. The Human Genome Project worked together to read every single word in that book and write them down so we could learn what makes us human.
The HGP advanced disease research by identifying genetic mutations linked to numerous conditions, fostering the development of targeted therapies. The HGP spurred biotechnological innovations, such as advanced DNA sequencing and bioinformatics tools, and established comprehensive genomic databases like GenBank. Some estimates put the economic impact of the Human Genome Project in the $1 Trillion range.
The first breakthrough for reviving Mammoths came in 2008 when scientists sequenced a large fraction of the mammoth genome from clumps of mammoth hair. Using CRISPR, they started copying mammoth DNA into living elephant cell cultures. Since then, they've made huge strides. Many genes have been successfully rewritten into Asian elephant cells, creating more mammoth-like cells with each edit. They've engineered mutations for mammoth hemoglobin, extra hair growth, fat production, and even climate adaptations like altered sodium ion channels in cell membranes. Why is this important? These advancements contribute to the fight against elephant endotheliotropic herpesviruses (EEHV)—a highly dangerous and often fatal condition affecting young Asian elephants and potentially leading to them becoming ‘endangered’
Here’s a great article from Asimov Press if you want a deeper explanation of the technological approach behind the research.
So…what’s happening now?
In 2021, the research was spun out from the Church lab at Harvard Medical School as Colossal Biosciences, with Dr. George Church and Ben Lamm at the helm. Already valued at over $1 billion, they are developing a de-extinction library of animals, slow biodiversity loss, and protect threatened species from extinction. Using this library of genomic sequences, they plan to rewild vital landscapes with de-extinct or hybrid species that are adapted to the climate challenges of today.
The man behind it all
If you haven’t yet heard of Dr. George Church - you’re certainly not going to forget him after this introduction. A molecular engineer, chemist, pioneer in synthetic biology, deep-tech entrepreneur, and a guest on the wildly popular Late Show with Stephen Colbert - Church has co-founded over 50 deeptech startups in his career, many of them being launched as spinoffs from the Church Lab at Harvard Medical School. George Church also helped initiate the Human Genome Project in 1984.
George Church is currently the Robert Winthrop Professor of Genetics at Harvard Medical School, a Professor of Health Sciences and Technology at Harvard University and Massachusetts Institute of Technology, and leads Synthetic Biology efforts for the Wyss Institute for Biologically Inspired Engineering at Harvard University. He also helped initiate the Human Genome Project (HGP) - the world’s largest collaborative biology project and certainly one of the most impactful projects in human history.
Today, we’re in conversation with him to further understand the potential of using biotech for biodiversity, gather his learnings from decades of commercializing deep technologies, and uncover where the puck is headed.
If you want to know George Church even more deeply, you can download his entire Genome (which he’s posted publicly) here. Just make sure you don’t clone him without permission.
With that, let’s dive in!
Interview with George Church
The Deep Tea: Why is biotech for biodiversity and conservation suddenly a thing now?
Dr. Church: This space is not new, but back in 1984 when I helped start the Human Genome Project, the main issue was the cost of DNA sequencing.. The big shift in DNA reading came with the move from electrophoretic sequencing, which dates back to around 1975, to next-gen sequencing, which I would say really took off around 2005, after about 30 years of development. It (next-gen DNA) sequencing was like a switch was flipped—we could now drop the price from $3 billion to $300. At $300, sequencing became economically viable and self-sustaining. It's akin to reaching the break-even point in controlled nuclear fusion, a kind of magical threshold that's almost within reach.
“It (next-gen DNA sequencing) was like a switch was flipped—we could now drop the price from $3 billion to $300. At $300, sequencing became economically viable and self-sustaining”
Around the $3,000 mark, we reached a tipping point where sequencing a small group of people in a country—like only 100 individuals—became feasible. Now, we can do comprehensive population genetics on every human and not just rely on sampling. This shift required us to rethink many lessons from the original Human Genome Project. Government grants supporting $1,000 genomes for next-gen sequencing also played a crucial role, opening the floodgates to sequencing every human and species, often with multiple examples of each.
So what we’re now able to do is population genetics on extinct species. For instance, there are 1,500 different specimens of the extinct passenger pigeon. Mammoths will likely have even greater representation due to the availability of high-quality, frozen DNA. There are tens of millions of frozen mammoths, which might be exposed as the permafrost melts, especially if we don't successfully address climate change.
The Deep Tea: When did you decide to take the tech out of the lab and commercialize it through Colossal?
Dr. Church: Around 2018, Ben (Co-Founder of Colossal) discovered my lab and was fascinated by all the projects. He realized that beyond achieving the Jurassic Park vision of his youth, our work could significantly aid conservation and technological development for veterinary and human health. I saw it the same way. Although I was working on these projects before meeting Ben, I didn't see their full potential and didn't seek grants or investors actively. In fact, I didn't bother to really write any grants,ask for any philanthropy or get any investors. I did get one philanthropic gift or maybe two, but in both cases, it wasn't because I was asking for it, it's because it just kind of showed up. And it was kind of the same way with Ben, he just showed up, and his vision was sort of a catalyst to push the research out of the lab.
The Deep Tea: That’s a really cool story. How do you balance running a research lab with being involved in startups? What are the strengths of each approach in advancing biotech for biodiversity?
Dr. Church: I typically avoid competing with my companies because they generally have 10-20 times more funding for projects. Companies often skip projects that don't fit investor timelines, while we can pursue them with grants and philanthropy, which are less restrictive. So for example, right now, our focus is on developing ways to create entire mammalian genomes from scratch, rather than just tweaking existing ones with multiplex editing. We've been working on genome synthesis for a long time, mostly with bacterial genomes up until now.
The big challenge is scaling up—moving from bacteria to mammals means we're talking about a thousand-fold increase in genome size. And to make it feasible, we also want to cut costs by a thousand-fold. Even synthesizing bacterial genomes is pricey, so bringing down costs while boosting accuracy and speed is key. This takes time and capital, without immediate returns, which a startup is not well-positioned for.
One of our projects is focused on developing artificial wombs, starting with experiments in mice. In larger biotech companies, similar outcomes are being achieved through cloning and using surrogates, as demonstrated in our previous work with pigs for organ transplants. Our novel research goal is to use these wombs to nurture any edited embryos and potentially bring them to term. Developing artificial wombs is complex, requiring deep knowledge in surgical techniques and developmental biology. Unlike projects that depend on computational or molecular methods, this effort demands direct biological experimentation and innovative approaches to tackle biological challenges and advance reproductive biology. This type of research is challenging to pursue within a large biotech company, which is usually focused on more extensive projects, and therefore its well positioned for a lab or academic environment.
The Deep Tea: Another thing we're curious about is the business side of de-extinction and what’s got VCs interested. What's the revenue model like for Colossal or similar organizations?
Dr. Church: Speaking about the Lab, we’ve made it clear that we're not aiming to profit much from conservation efforts. This is similar to how CRISPR is made available to the academic research community, more or less for free. However, we still saw business opportunities through spin-off companies like Editas and Egenesis, which focused on gene therapies and organ transplants.
“We still saw business opportunities through spin-off companies…For Colossal, they’ve told investors that their focus is on technology spinoffs”
For Colossal, they’ve told investors that their focus is on technology spinoffs. So far, they've spun off one company called FormBio, with more expected soon. These spinoffs could involve various technologies—some computational, some biotech—as well as educational and entertainment components. Ben Meserec's book on Mammoths, "Woolly", has inspired a $160 million project by 20th Century Fox to make it into a movie, and that highlights broad interest and potential in these ventures beyond conservation.
The Deep Tea: You earlier spoke about the reduction in cost of sequencing - how does this translate into a business model? Secondly, given the high costs involved, how can ecosystem players such as governments/ investors better support startups in this space to expedite their path to achieving their core business models, especially those lacking initial infrastructure support like Colossal had?
Dr. Church: The great thing about exponential technologies is how quickly costs can drop and keep decreasing. We've seen this with electronics, the internet, and now with reading and writing DNA from $3 billion to $300. Imagine if out of 100 families, 3% are at risk of million-dollar diseases. You can sequence all of them, even if only 3% benefit—it's still millions of dollars generated that can then be reinvested and scaled to sequence the next 1000 families, and benefit even more people. The break-even point for this kind of DNA Analysis was around $3,000, with a tenfold return at a $300 cost. Governments or big healthcare providers could benefit hugely from scaling this, but few are fully on board yet.
Developing this tech doesn't require a lab in every country. We make it widely available for academics and non-academics through patents and licensing. Innovation doesn't need a big budget; every few years, new breakthroughs can level the playing field globally.
Our contributions to next-gen sequencing and CRISPR cost about $2 million each. We transferred them to companies fully functional, not just academic papers. This approach encourages innovation everywhere, from any country, at a low cost, while deterring copycats.
The Deep Tea: You've attracted a diverse mix of investors, from agri-bioscience VCs to even Paris Hilton. I'm curious about the VC perspective. Typically, VCs aim to exit in 8 to 12 years. How does Colossal, with its big mission of de-extinction, fit their funding timelines and expectations?
Dr. Church: Well, so there's different VCs, all with different timelines and theses. Stereotyping a little bit, on the West Coast, quick returns are common with internet startups that can become hits almost immediately with the right coding and marketing, offering a three-year ROI expectation. At the other extreme is the East Coast of the United States, where people are investing in something that might take eight years of research and development, and then another eight to 10 years to get through FDA approval. And then another period of time where you have to do the marketing and sales. So basically at the end of 18 years, you're kind of where the little internet startup was when it started.
So, think of Colossal like this: If you're in the drug market, it can take 18 years to see results, which is reasonable, especially for big goals like solving climate change or saving species. But if your focus is on our technology, the important thing to notice we've already spun off a company from Colossal within a year.
The Deep Tea: Let’s talk about some of your learnings from commercializing deep technologies. First of all, how did this prolific entrepreneurial journey start?
Dr. Church: My labs first brush with startup fundraising was when a post-doc in my lab, Francois Vigneault decided to spin off an immunology company, Abvitro. And that was, it was probably in 2008-ish. And he started it in his bathroom in his home, where he had a baby and everything. It was even lower tech than the typical garage lab stereotype! That startup got acquired very quickly, in about three years or so, and that in turn was acquired by Celgene which produced a number of important immunotherapies. Francois went on to do other things, and we’re currently working on a new startup named Shape Therapeutics.
But this was a key transition - after this experience with Francois and Juno Therapeutics, a lot of my postdocs would look at that story and say, hey, we could do that. And from that Lujan formed E-genesis, and then we formed Editas, and it just snowballed from there. Now, everything we think of in the lab generates one or two or as many as six companies.
“Now, everything we think of in the lab generates one or two or as many as six companies”
The Deep Tea: What do you think are common pitfalls with deeptech investing and commercialization that VCs often miss?
Dr. Church: Often when there’s a big, shiny new technology, VC’s can sometimes miss the bigger picture. Let me give you an example - consider machine learning applied to protein engineering. Uniref, a 2019 paper that Ethan Alley was first author on, was the first machine learning model for protein design. And at the time, everybody was distracted by machine learning for protein structure. And we did it instead for protein design at the sequence level, not 3D structure.
And so VCs didn't see what was happening under their nose, because they were distracted by ML for protein structure and this new shiny object for 3D structure called AlphaFold. Meanwhile though, our efforts in ML for protein design spun off six companies, which are Manifold, Dyno, Nabla, JURA, Shape, and Patch (acquired by Ginkgo Bioworks). And they were all quite different from each other, but they all had in common that they saw the power of not just the machine learning, but combining it with multiplex libraries, which we had introduced in around 2004 Nature paper, but nobody really, nobody other than our collaborators had caught on to it yet.
And so we had this big grace period where we were able to develop that. And that combination, machine learning plus multiplex libraries was just unstoppable. I think we still haven't quite seen the full vision of where it's going, that combination. So I think that was a weakness from the VC side, falling for the hype and missing the bigger picture of what technologies like machine learning enable in biotech.
“…that was a weakness from the VC side, falling for the hype and missing the bigger picture of what technologies like machine learning enable in biotech”
The Deep Tea: Finally, just for fun, what's your take on the next big revolution? Any underrated dark horse technology you're excited about for the next 10, 20, or even 30 years?
Dr. Church: The thing I'm most excited about, and maybe three that are under appreciated – Number one is genetic counseling . VCs are throwing a lot more money at gene therapy than at genetic counseling, even though genetic counseling solves 99% of what you can do with gene therapy. And it does it for 10,000 times less money. So I think that's number one. That's a great opportunity, and it's not like there's any less money there. Just because it's 10,000 times cheaper doesn't mean there's less money there. It's the same argument for next-gen sequencing. Next-gen sequencing is 20 million times cheaper than previous sequencing methods. But people are making more money with next-gen sequencing than they were with first-gen, because it's a bigger market. I think that genetic counseling is completely ignored.
Second thing is developmental biology and aging. So even though there's a great increase in aging-related research and investment - we’re missing the point, there’s a trillion dollars for every year of youthful life we gain. There’s a lot ways you can recover that.
Third thing is “natural computing” in which poor (silicon digital) simulations of complex problems (such as targeting ad avoiding off targets in a complex body are replaced with perfect actual tests with up to trillions of shots on goal and machine learning to guide cycles of optimization.
Conclusion
In conclusion, we believe that the intersection of biotechnology and biodiversity offers transformative potential beyond the realms of de-extinction and conservation. As we have seen, the innovative endeavors by Colossal reflect a broader vision, extending to the enhancement of human health and longevity. Addressing issues such as herpes in elephants through gene editing has shown promise with humans too, highlighting the intricate link between animal health, human health and holistic conservation efforts. By leveraging biotech solutions, we can create more resilient ecosystems and improve the quality of life for both wildlife and humans, paving the way for a future where the health of our planet and its inhabitants are intricately and sustainably connected.
Have some thoughts? We’d love to hear from you